Direct synthesis of acetic acid from carbon dioxide and methane over Cu-modulated BEA, MFI, MOR and TON zeolites: a density functional theory study†
Abstract
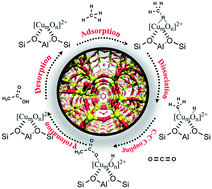
Direct synthesis of acetic acid from carbon dioxide and methane is considered a green and atom-economic process, which can rationally utilize two gases contributing mostly to the greenhouse effect; however, this process faces a tremendous challenge of finding an efficient catalyst due to the chemical inertness of methane and carbon dioxide. Herein, the catalytic conversion of carbon dioxide and methane to acetic acid over Cu-modulated BEA, MFI, MOR and TON zeolites, including the adsorption of CH4, dissociation of C–H bonds, C–C bond coupling, protonation of acetate, and desorption of acetic acid, was investigated by using the periodic density functional theory (DFT) calculation. The results indicate that the catalytic activity of Cu-modulated zeolites is closely related to the structure of both active copper species and zeolite pore channels. The binuclear copper-oxo cluster ([CuOCu]2+) in the three-dimensional BEA and MFI zeolites and trinuclear copper-oxo cluster ([Cu3(μ-O)3]2+) in the one-dimensional MOR and TON zeolites are catalytically active in the direct conversion of carbon dioxide and methane to acetic acid, in comparison with the mononuclear copper ([Cu]2+) species. In particular, the MOR zeolite carrying [Cu3(μ-O)3]2+ clusters ([Cu3(μ-O)3]-MOR) exhibits superior catalytic activity in the production of acetic acid with a low apparent activation energy (57 kJ mol−1). The insights shown in this work should be helpful in clarifying the catalysis of Cu-modulated zeolites and the reaction mechanism of producing acetic acid from carbon dioxide and methane, which can provide some clues for the design of efficient catalysts in the conversion of carbon dioxide and methane.
- This article is part of the themed collection: 2019 Catalysis Science & Technology HOT Articles


 Please wait while we load your content...
Please wait while we load your content...