Ultrafine PdOx nanoparticles on spinel oxides by galvanic displacement for catalytic combustion of methane†
Abstract
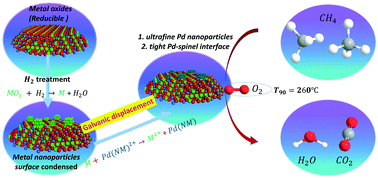
High light-off temperature and poor water-tolerance at low temperature are the main drawbacks of catalysts for catalytic methane combustion. Herein, the galvanic displacement method was adopted to obtain ultrafine PdOx nanoparticles (∼1 nm) on NiCo2O4 and build a tight Pd–NiCo2O4 interface. As a comparison, additional reference samples were prepared by conventional synthetic methods. The synthesized catalysts were characterized by XRD, TEM, XPS, H2-TPR, CH4-TPR, CO2-TPD and in situ DRIFT techniques. It is found that the catalyst obtained by galvanic displacement has more Pd4+ and oxygen vacancies. The high oxidation state of Pd is more conducive to the activation of methane, and a large number of oxygen vacancies can promote the catalytic reaction rate. Thus, this catalyst displays outstanding catalytic activity with T90 = 260 °C and excellent water-tolerance below 300 °C. In addition, a simple model was built based on the obtained results to understand the catalytic mechanism and provide a universal strategy for rational design of catalysts for low temperature complete oxidation of methane.


 Please wait while we load your content...
Please wait while we load your content...