Transformations of FCC catalysts and carbonaceous deposits during repeated reaction-regeneration cycles†
Abstract
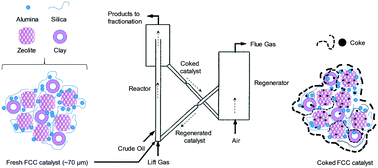
Transformation of an industrial zeolite-based fluid catalytic cracking (FCC) catalyst and its coke deposits during regeneration following FCC reactions of a representative refinery stream (a 1 : 1 weight basis mixture of light cycle oil and heavy gas oil) are investigated. The coked catalysts are regenerated on stream, between reaction cycles, in a regenerator maintained at 700 °C. Coked samples after 1, 22 and 45 reaction cycles (FCC-1, FCC-22 and FCC-45) are collected and analyzed using X-ray photoelectron spectroscopy (XPS), in situ Raman spectroscopy, X-ray diffraction (XRD), nitrogen and oxygen adsorption–desorption isotherms, scanning electron microscopy (SEM) and 27Al magic angle spinning nuclear magnetic resonance (MAS NMR) spectroscopy. Our results indicate that the amount of coke decreased, and the coke concentrated preferentially on the surface of the catalyst particles as the catalyst is subjected to an increasing number of reaction-regeneration cycles. A decrease in the micropore volume and total acidity of the catalyst is observed due to pore blockage by coke and the damage to the crystalline structure of the catalyst induced by cycling. The most significant catalyst deactivation is observed during the first reaction step. The increased number of reaction cycles and thermal aging also change the structural order of the graphitic species or the morphology of the coke, producing harder coke that is more difficult to oxidize.


 Please wait while we load your content...
Please wait while we load your content...