Biomimetic catalytic oxidative coupling of thiols using thiolate-bridged dinuclear metal complexes containing iron in water under mild conditions†
Abstract
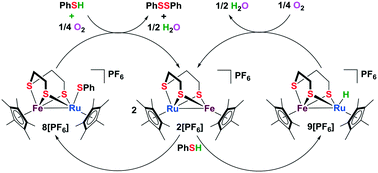
A green and efficient approach to disulfides via oxidative coupling of thiols was developed by adopting a biomimetic thiolate-bridged iron–ruthenium complex as the catalyst. Using environmentally friendly oxygen as the oxidant, a wide range of thiols including biologically important molecules can be smoothly converted into corresponding disulfides in water. Notably, two potential intermediate species were successfully isolated and unambiguously characterized, which is essential to reveal the detailed mechanism of this transformation. This catalytic system represents a rare and desired heteronuclear bimetallic scaffold for understanding the biological process of S–S bond formation from the viewpoint of bioinspired catalysis.


 Please wait while we load your content...
Please wait while we load your content...