Low-loading Pt nanoparticles embedded on Ni, N-doped carbon as superior electrocatalysts for oxygen reduction†
Abstract
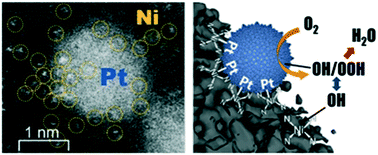
As one of the efficient and classic nanoscale catalysts, Pt nanoparticles play predominant roles in multiple energy conversion systems, especially for electrochemical devices involving the oxygen reduction reaction (ORR). It is important to develop a scalable method for synthesis of more efficient Pt-based electrocatalysts with higher activity and stability. In this work, a low-loading Pt-based electrocatalyst (8.0 wt%) is fabricated by the galvanic replacement reaction, presenting well-dispersed Pt nanoparticles adjacent to atomic Ni–N–C complexes (Pt@NiNC). Due to the synergetic effect associated with the nanoscale/atomic scale joint active sites and the strong metal–support interaction, the resulting Pt@NiNC hybrid exhibits better ORR performance and higher mass activity than the benchmark Pt/C, as well as enhanced electrochemical stability. This research not only opens a new route to develop heterogeneous catalysts with multi-scale joint sites but also provides bright prospects for high-performance and low-cost energy conversion and storage.


 Please wait while we load your content...
Please wait while we load your content...