Understanding the molecular properties of the E1 subunit (SucA) of α-ketoglutarate dehydrogenase complex from Vibrio vulnificus for the enantioselective ligation of acetaldehydes into (R)-acetoin†
Abstract
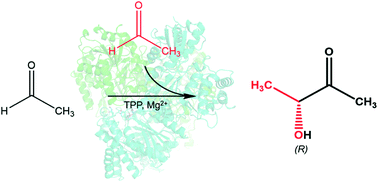
Escherichia coli SucA, a decarboxylating E1 component of the α-ketoglutarate dehydrogenase complex, has been reported to possess another catalytic activity in carboligating two acetaldehyde molecules to form acetoin in a thiamine diphosphate (ThDP)-dependent manner. Here, we examined acetoin formation activity from acetaldehyde using SucAs from various organisms, as well as the Zymomonas pyruvate decarboxylase (ZmPDC) and the formaldehyde-ligating formolase. Among the studied SucA enzymes, Vibrio vulnificus SucA (VvSucA) exhibited the highest ligating activity against acetaldehyde with an excellent enantioselectivity of the product in contrast to ZmPDC and formolase of poor enantioselectivity. The revealed VvSucA structure shows a dimeric assembly with the bound ThDP within the active sites at the two-subunit interface. Non-covalently bound glycolaldehyde molecules suggest the two substrate-binding sites within the carboligating active site for acetaldehyde. Structure-guided mutants reveal residues critical for regiospecificity of VvSucA. The obtained biochemical and structural data help to understand the high enantioselective ligation of two acetaldehyde molecules by VvSucA.


 Please wait while we load your content...
Please wait while we load your content...