Switching acidic and basic catalysis through supramolecular functionalization in a porous 3D covalent imine-based material†
Abstract
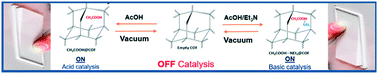
Non-covalent inclusion of small acid and base molecules (CH3COOH and NEt3) in an imine structure based on micrometre COF-300 crystals and amorphous nanoparticles results in the facile modulation of their acid–base properties. Such a strategy results in the triggering of acidic/basic catalytic activity of the otherwise inactive materials towards ring-opening epoxide and Knoevenagel condensation reactions. For both reactions, amorphous nanoparticles are better catalysts than micrometre crystals as they exhibit a higher external surface area. The found activities and stability of this supramolecular functionalization are modulated by confinement effects, which are rationalized with the help of DFT calculations. All results obtained suggest that the reactions catalysed by these functionalized materials occur confined to pores closer to the material surface, which makes size-discrimination phenomena possible, and explains the major activity of the nanoparticulated material.


 Please wait while we load your content...
Please wait while we load your content...