Self-supported CoFe LDH/Co0.85Se nanosheet arrays as efficient electrocatalysts for the oxygen evolution reaction†
Abstract
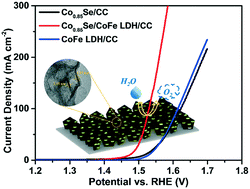
The exploration of high-performance, cost-effective, and stable electrocatalysts for the oxygen evolution reaction (OER) is crucial for future energy storage and conversion technologies. Cobalt-based hydroxides and oxides have been considered as ideal OER electrocatalysts and have been intensively studied; however, their catalytic activity is still unsatisfactory. It is known that the interfacial electron and structure engineering of composite electrocatalysts often results in synergistically enhanced catalytic performance. Herein, a self-supported binary heterogeneous catalyst composed of CoFe LDH/Co0.85Se nanosheet arrays vertically grown on carbon cloth has been reported. The heterostructure electrocatalyst exhibits excellent activity for the OER in alkaline electrolyte, requiring an overpotential of only 241 mV at a current density of 10 mA cm−2 with a small Tafel slope of 48 mV dec−1. These performances are significantly better than those of the bare CoFe LDH and Co0.85Se catalysts. Moreover, the composite electrodes exhibit outstanding durability within 40 h of testing. Our further electrochemical results show that the excellent activities of the heterostructure electrodes should be ascribed to the interfacial coupling effect, increased number of activity sites, accelerated electron transfer, and strong structural stability. This work paves an interesting route for boosting the OER performance of oxide-based electrocatalysts by the regulation of electronic structure through introducing non oxide electrocatalysts for various applications.


 Please wait while we load your content...
Please wait while we load your content...