Incorporating silicon carbide nanoparticles into Al2O3@Al to achieve an efficient support for Co-based catalysts to boost their catalytic performance towards Fischer–Tropsch synthesis†
Abstract
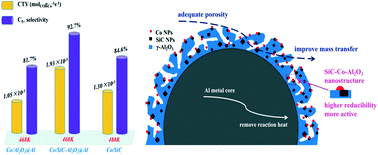
Herein, the shell–core-structured SiC–Al2O3@Al composite consisting of an Al metal core and a SiC–Al2O3 hybrid shell was synthesized by a straightforward preparation method. Compared with Al2O3@Al composites, the SiC-modified samples possess a meso–macroporous structure, high pore volume, wide pore size, and a special surface where active and inert components coexist. The ratio of inert to active components on the composite surface could be modulated by controlling the amount of SiC nanoparticles (NPs). Upon the decoration of SiC NPs, the degree of reduction of cobalt oxides and the density of active sites (Co0) increased significantly. Moreover, the SiC-modified catalysts exhibited lower apparent activation energy (Ea) and higher intrinsic reaction rate (TOF) mainly due to the electron-rich properties of Co NPs in the modified samples. Accordingly, the cobalt-weight normalized FT activity (CTY) of the SiC-modified catalysts increased by a factor of about 1.6–2.0. Moreover, the catalyst modified by the SiC NPs showed lower selectivity towards light hydrocarbons (CH4 and C2–C4) but higher selectivity towards long-chain hydrocarbons (C5+). When the mSiC/mAl ratio was 15%, the CoAC-15 catalyst showed maximum density of active sites and highest TOF, leading to highest activity and C5+ selectivity (approximately 92%). The synergistic effect of the SiC–Al2O3 hybrid shell and the Al metal core was beneficial for the catalytic properties of the Co/SiC–Al2O3@Al catalysts. The binary SiC–Al2O3 nanostructure endowed the Co NPs with electron-rich characteristics to enhance the CO activation; moreover, the special surface where the active and inert components coexisted improved the reducibility of cobalt oxides, and the abundant porosity of the hybrid shell facilitated the transfer of reactants and products. In addition, the existence of the Al metal core improved heat transfer in the reaction.


 Please wait while we load your content...
Please wait while we load your content...