Synthesis of water-soluble gold–aryl nanoparticles with distinct catalytic performance in the reduction of the environmental pollutant 4-nitrophenol†
Abstract
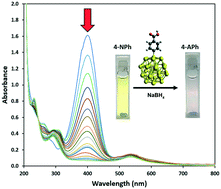
Catalysis under harsh environmental conditions requires robust nanoparticles that can resist leaching of the organic shell and possess significant resistance to aggregation. Robust gold core-carbon shell gold–aryl nanoparticles (AuNPs–COOH) were fabricated by mild reduction of the water-soluble aryldiazonium salt [HOOC-4–C6H4N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N]AuCl4, and were fully characterized in solution and solid state. The nanoparticles showed high stability in the presence of 0.01–1.00 M NaCl salt, acidic and basic pH values (1–13) and moderate temperatures (20–90 °C). DFT calculations of the optimized model system Au38–C6H4–COOH show an Au–C (aryl) distance of 2.04 Å, which is related to a binding energy of −59.2 kcal mol−1. The 4-nitrophenol (4-NPh) reduction model was used to study the viability of AuNPs–COOH as a catalyst. Nitrophenols are among the most common organic pollutants in industrial and agricultural wastewaters due to their toxicity, anthropogenic and inhibitory nature. The AuNPs–COOH show high catalytic activity, where the reduction of 80 μM 4-NPh was complete in less than five minutes with a high kapp (2.26 × 10−2 s−1) and a relatively low Ea (25 kJ mol−1) compared to literature values. Catalytic activity decreases with subsequent cycles of the reaction, along with a decrease in intensity and red shift in the LSPR band, and an increase in aggregation of nanoparticles in the TEM following each reaction cycle.
N]AuCl4, and were fully characterized in solution and solid state. The nanoparticles showed high stability in the presence of 0.01–1.00 M NaCl salt, acidic and basic pH values (1–13) and moderate temperatures (20–90 °C). DFT calculations of the optimized model system Au38–C6H4–COOH show an Au–C (aryl) distance of 2.04 Å, which is related to a binding energy of −59.2 kcal mol−1. The 4-nitrophenol (4-NPh) reduction model was used to study the viability of AuNPs–COOH as a catalyst. Nitrophenols are among the most common organic pollutants in industrial and agricultural wastewaters due to their toxicity, anthropogenic and inhibitory nature. The AuNPs–COOH show high catalytic activity, where the reduction of 80 μM 4-NPh was complete in less than five minutes with a high kapp (2.26 × 10−2 s−1) and a relatively low Ea (25 kJ mol−1) compared to literature values. Catalytic activity decreases with subsequent cycles of the reaction, along with a decrease in intensity and red shift in the LSPR band, and an increase in aggregation of nanoparticles in the TEM following each reaction cycle.


 Please wait while we load your content...
Please wait while we load your content...