Can Hammett indicators accurately measure the acidity of zeolite catalysts with confined space? Insights into the mechanism of coloration†
Abstract
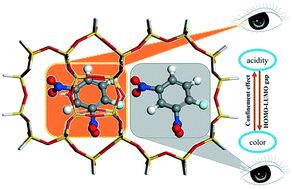
The acidic properties of zeolite catalysts play a crucial role in governing catalytic performances, which makes the acidity characterization an important subject in the field of zeolite catalysis. So far, Hammett titration has been a conventional method to determine the acidic strength of liquid and solid acids, however, the reliability of this titration method to measure the acidity of acidic sites inside zeolites is in debate. It is necessary to unveil the origin of the coloration of Hammett indicators adsorbed inside zeolite channels and cages. Herein, by employing an advanced computational method, the adsorbed structures and electronic properties of a series of Hammett indicators with various pKa (from −0.29 to −14.52) values have been investigated, concerning the interaction with the Brønsted acidic sites inside H-ZSM-5, H-MOR and H-Y zeolites. On the basis of the theoretical results, the intimate relationship between the coloration (traditionally corresponding to the protonation of Hammett indicators) and HOMO–LUMO gap has been established by DFT and time-dependent (TD) DFT calculations for the first time. Furthermore, not only the Brønsted acidity, but also the pore confinement derived from the zeolite framework could also render the coloration by effectively reducing the HOMO–LUMO gaps, which can be differentiated by the diverse shapes and sizes of zeolite cages. Therefore, one should be cautious in the utilization of Hammett titration for the acidity determination of porous zeolite catalysts and other solid acids, and other more accurate approaches are needed in the future.
- This article is part of the themed collection: 2019 Catalysis Science & Technology HOT Articles


 Please wait while we load your content...
Please wait while we load your content...