Ce/MgAl mixed oxides derived from hydrotalcite LDH precursors as highly efficient catalysts for ketonization of carboxylic acid†
Abstract
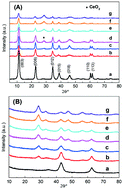
Ketonization of carboxylic acids provides an attractive way for upgrading biomass feedstocks into biofuels. Here, a series of Mg–Al hydrotalcite (HT) catalysts doped with different Ce contents were prepared and used for the ketonization of propionic acid. Mg3Al0.9Ce0.1 displayed the highest ketonization activity (90.6% conversion of propionic acid) under mild reaction conditions (350 °C). By applying SEM, TEM, H2-TPR, CO2/NH3-TPD, XPS, etc., the mechanism for the improvement of catalytic activity and stability was obtained. When the doping content of Ce is low, such as in Mg3Al0.9Ce0.1, cerium oxide is uniformly dispersed on the HT support. At the same time, the strong interaction between the CeO2 species and Mg–Al hydrotalcite led to the improvement of redox properties and modified acid–base sites, which is beneficial for the ketonization reaction. However, the structure of the HT support is destroyed by the agglomerated cerium oxide when a more doped Ce is employed, and the specific surface area and the surface Ce3+ ratio are remarkably lowered, thereby causing a significant decrease in the ketonization activity. By using the temperature programmed surface reaction (TPSR) technique, a deeper insight into the mechanism of reaction pathways over different catalysts is gained. The significant improvement in the reaction stability of the ketonization reaction over Mg3Al0.9Ce0.1 as compared to unmodified catalysts could be ascribed to the strengthening of the M–O bond of the catalyst after the introduction of Ce.


 Please wait while we load your content...
Please wait while we load your content...