Combined halide-free Cu-based catalysts with triple functions for heterogeneous conversion of methanol into methyl acetate†
Abstract
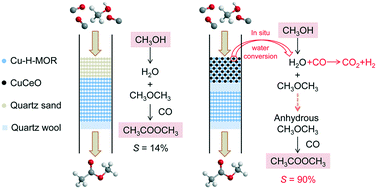
Heterogeneous conversion of methanol to methyl acetate (MA) through halide-free catalysis is meaningful from academic and industrial perspectives. In this study, we developed a combined halide-free Cu-based catalyst system composed of Cu-H-MOR + CuCeO and Cu-H-MOR (labeled Cu-H-MOR + CuCeO‖Cu-H-MOR) for the heterogeneous conversion of methanol into MA in the presence of CO. Cu-H-MOR catalyzed the dehydration of methanol to dimethyl ether (DME) and subsequent carbonylation of DME to MA. CuCeO converted water from methanol dehydration into H2 and CO2 through the water-gas shift reaction (WGSR) at relatively low temperatures. The WGSR catalyst is critical to the conversion of methanol to MA in the presence of CO through halide-free catalysis. This process not only produced H2 but also eliminated significantly the influence of water on the carbonylation of DME. Results indicated that the selectivity to MA was only 14% without the WGSR catalyst but reached 90% with the WGSR catalyst. Thermal gravimetric analysis showed that the selectivity to MA began to decline gradually after the reaction on stream for 18 h because of the coke formation in the catalyst pores. The coke on the catalyst could be completely removed simply by burning without destroying the structure of the combined catalyst. The combined catalyst system Cu-H-MOR + CuCeO‖Cu-H-MOR could be recycled at least five times with negligible loss in activity and MA selectivity.


 Please wait while we load your content...
Please wait while we load your content...