In situ synthesis of FeP-decorated Ti–Fe2O3: an effective strategy to improve the interfacial charge transfer in the photoelectrochemical water oxidation reaction†
Abstract
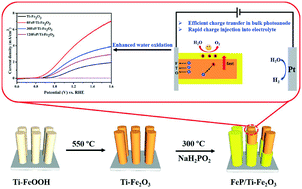
Modifying inexpensive cocatalysts on semiconductors to accelerate the charge transfer at the photoanode/electrolyte interface, which possess good charge transfer in the bulk photoanode simultaneously, is priority among priorities in constructing low-cost and efficient PEC solar energy conversion devices. However, it is often difficult to satisfy outstanding charge transfer both in the bulk and at the photoanode/electrolyte interface when integrating cocatalysts with semiconductors. In our work, we synthesized FeP-decorated Ti–Fe2O3in situ through simple low-temperature phosphidation, which possessed good interfacial contact between Ti–Fe2O3 and FeP. The results demonstrated that the in situ deposition of FeP not only reduced the charge transfer resistance in the bulk photoanode by improving the electrical conductivity, but also accelerated the charge transfer at the photoanode/electrolyte interface. Based on the above advantages, the novel FeP/Ti–Fe2O3 exhibited splendid photocurrent density as high as 3.9 mA cm−2 at 1.23 V vs. RHE; meanwhile, the onset potential of FeP/Ti–Fe2O3 shifted cathodically to 0.88 V vs. RHE in comparison with that of Ti–Fe2O3 (0.98 V vs. RHE). This work paves a simple path to construct a cocatalyst-modified photoanode with excellent interfacial charge transfer and offers a low-cost and promising cocatalyst that can be applied in the photoelectrochemical water oxidation reaction.


 Please wait while we load your content...
Please wait while we load your content...