Visible light-driven enhanced CO2 reduction by water over Cu modified S-doped g-C3N4†
Abstract
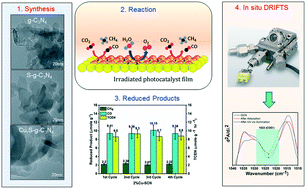
We synthesized Cu modified S-doped g-C3N4 thin sheets using calcination followed by a wet-impregnation method. Herein, the photocatalytic performance of the as-synthesized sheets was systematically studied for gas-phase CO2 reduction using water at room temperature under visible light illumination. The optimum photocatalyst, 2% Cu-SCN, illustrated the best production rates of CO and CH4, approximately 3 and 7 times higher, respectively, than those of pristine g-C3N4. Stability testing also revealed the prolonged lifetime of 2% Cu-SCN. Characterization results confirmed that Cu existed in Cu0, Cu+, and Cu2+ oxidation states. Also, Cu was efficient at extending the absorption wavelength range and reducing recombination events, partially owing to surface plasmon resonance effects. In situ diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) studies suggested that COO− and HCHO are the active intermediates for CO formation. Moreover, bicarbonate species had a higher absorbance value on the Cu-modified sheets, and could be an intermediate for CH4 production. The findings revealed that Cu0/Cu+ played a pivotal role in increasing methane selectivity. The present work thus successfully demonstrated the synergistic use of both a metal and non-metal on a host photocatalyst to be a promising strategy for efficient CO2 reduction.
- This article is part of the themed collection: 2019 Catalysis Science & Technology HOT Articles


 Please wait while we load your content...
Please wait while we load your content...