Anchoring ultrafine PtNi nanoparticles on N-doped graphene for highly efficient hydrogen evolution reaction†
Abstract
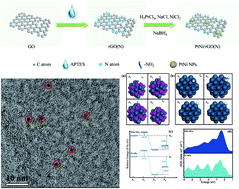
The exploration of high-efficiency electrocatalysts for the hydrogen evolution reaction (HER) is of great significance for sustainable energy conversion applications, yet it remains a grand challenge. Herein, a facile and rapid strategy to synthesize ultrafine PtNi nanoparticles (NPs) anchored on N-doped graphene (rGO(N)) (labeled as PtNi/rGO(N)) at room temperature is demonstrated. (3-Aminopropyl) triethoxysilane (APTES) is selected as an effective nitrogen source to form two kinds of nitrogen (doping N and amine N) simultaneously for fabricating a rGO(N) matrix during the chemical reduction process. Benefiting from the bimetallic synergistic effect and strong metal–support interactions, this composite is expected to accelerate H+ adsorption and H2 desorption and reduce the transport resistance of electrons and hydrogen intermediates. As a consequence, the PtNi/rGO(N) with ultralow Pt loading amount (1.2 μg per electrode area (cm2)) exhibits extraordinary catalytic activity with a small overpotential of 98 mV at a current density of 10 mA μgPt−1 and an exceptional Tafel slope of 42.7 mV dec−1 for HER, exceeding the incumbent commercial Pt/C catalyst. Moreover, the PtNi/rGO(N) also displays excellent stability with negligible current degradation during a continuous operation for 20 h. The present work would be proposed as an elegant platform toward the exploration of efficient HER electrocatalysts for various renewable energy conversion applications.


 Please wait while we load your content...
Please wait while we load your content...