Highly selective production of renewable p-xylene from bio-based 2,5-dimethylfuran and ethylene over Al-modified H-Beta zeolites†
Abstract
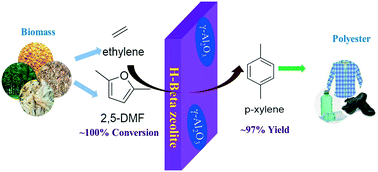
Highly selective synthesis of renewable p-xylene (PX) from bio-based 2,5-dimethylfuran (DMF) and ethylene was successfully realized on H-Beta zeolite catalysts with further Al-modifications. The influences of different doping approaches on their reaction performances and coke deposition were systematically investigated. Solid-state NMR and SEM-EDS characterization indicates that the Al containing precursors have significant effects on the aluminum location and catalyst acidity. For H-Beta zeolite treated with aluminum nitrate, the contents of framework Al and Brønsted acid sites increase obviously, causing the formation of more hard coke on the used catalyst as shown in 13C CP/MAS NMR and TPO profiles, while for H-Beta zeolite modified with aluminum isopropoxide, more non-framework Al are formed in the pores of H-Beta, leading to severer soft and hard coke deposition. However, H-Beta zeolite after further doping with alumina shows excellent and durable performances with a PX yield of 97% at a 2,5-DMF conversion of ∼100%. It also has good stability and regeneration capability. Most importantly, the coke content decreases from 12 to 7 wt% on the spent H-Beta-alumina catalyst with mainly soft carbonaceous species due to its appropriate Brønsted/Lewis acid ratio. This work provides a deeper understanding of the role of B/L ratio in the sustainable conversion of 2,5-DMF and ethylene to p-xylene, paving the way for the synthesis of bulky chemicals from biomass feedstocks.


 Please wait while we load your content...
Please wait while we load your content...