Probing the active sites of newly predicted stable Janus scandium dichalcogenides for photocatalytic water-splitting†
Abstract
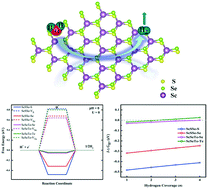
The Janus structures of transition metal dichalcogenides with intrinsic dipoles have recently drawn attention as efficient candidates in the class of non-precious metal photocatalysts for water splitting. Here, we have proposed Janus ScXY (X/Y = S, Se, or Te, X ≠ Y) monolayers as promising catalysts for the hydrogen evolution reaction (HER). We have thoroughly investigated the structural stabilities of the ScXY monolayers, as a primary step to understand the splitting mechanism more accurately. It has been observed that Janus ScSSe and ScSeTe monolayers are dynamically, thermally and mechanically stable, whereas the ScSTe monolayer is dynamically unstable. Moreover, based on hybrid functional (HSE06) calculations, these stable ScSSe and ScSeTe monolayers are semiconductors with direct band gaps of 1.810 and 0.921 eV, respectively. Considering spin–orbit coupling on top of the HSE06 calculations results in an insignificant change in the band gaps. The steady optical responses of Janus ScSSe and ScSeTe, respectively, in the visible and near IR spectra demonstrate their photocatalytic activities in the respective regions. Interestingly, we have observed excellent performance shown by the ScSeTe monolayer, where both surfaces are catalytically active, and maintain close to thermoneutral conditions with near-zero adsorption free energy at different H* coverages. Comparatively, the ScSSe monolayer shows poor performance for the HER due to strong bonds with the adsorbed hydrogen. Also, it has been confirmed that even the application of ±2% strain on the ScSSe monolayer does not enhance its HER activity. Our theoretical predictions demonstrate that out of all the Janus ScXY monolayers studied here, ScSeTe is the most active catalyst for the HER to efficiently split water, which certainly could make it worth performing further experimental investigations.


 Please wait while we load your content...
Please wait while we load your content...