Effect of initial Si/Al ratios on the performance of low crystallinity Hβ-x zeolite supported NiMo carbide catalysts for aromatics hydrogenation†
Abstract
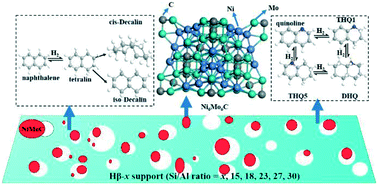
A series of mesoporous Hβ zeolites with low crystallinity and various Si/Al ratios were prepared using different Si/Al ratios in the reactants. Then, NiMoC/Hβ-x catalysts (x = 15, 18, 23, 27 and 30) were prepared using a co-impregnation method followed by temperature-programmed reduction under a H2/CH4 atmosphere. The effects of the initial Si/Al molar ratio on the chemical compositions, textural properties, acidities and morphologies of these materials were investigated by X-ray diffraction (XRD), scanning electron microscopy (SEM), X-ray fluorescence (XRF), N2 adsorption–desorption, temperature-programmed ammonia desorption (NH3-TPD), FTIR spectroscopy of adsorbed pyridine (Py-FTIR), H2-temperature programmed reduction (H2-TPR), high resolution transmission electron microscopy (HRTEM) and X-ray photoelectron spectroscopy (XPS). The catalytic performance towards the hydrogenation of naphthalene was tested with and without quinoline in a fixed-bed reactor, and then evaluated by hydrogenation of aromatics from low temperature coal tar (LTCT). The results showed that the total acidity, the number of Brønsted acid sites and Lewis acid sites, and the molar ratio of Brønsted to Lewis acid sites (B/L) decreased with increasing initial Si/Al ratio for the pure Hβ-x zeolites and their corresponding supported NiMoC catalysts. The NiMoC/Hβ-18 catalyst had the best catalytic activity among the prepared catalysts and a strong tolerance to heteroatom compounds in the hydrogenation of aromatic compounds. It could be concluded that the total acidity of the catalyst and the dispersion of the active metal served as crucial factors that determined the catalytic performance toward aromatics hydrogenation.


 Please wait while we load your content...
Please wait while we load your content...