Ultrafine Au nanoparticles anchored on Bi2MoO6 with abundant surface oxygen vacancies for efficient oxygen molecule activation†
Abstract
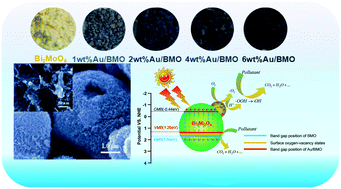
Oxygen molecule activation is one of the most important processes to generate oxygen-containing active free radicals for organic pollutant photocatalytic decomposition. Herein, we demonstrated ultrafine Au nanoparticles (NPs) anchored on porous Bi2MoO6 (BMO) microspheres with rich surface oxygen vacancies (SOVs) planted via a simple chemical reduction–deposition method, which could effectively enhance the separation of photogenerated carriers for oxygen molecule activation, and thus lead to a more efficient photocatalytic decomposition ability of the phenol and dye. The sample (4.0% Au/Bi2MoO6) exhibited a remarkable photocatalytic performance for phenol, which is 15 times higher than BMO. The systematic studies indicate that the excellent photocatalytic activity of Au/Bi2MoO6 should be ascribed predominantly to the synergistic effect between SOVs, Au NPs and BMO. Both SOV and Au NP surface plasmonic resonance (SPR) can not only improve the separation and migration of the photogenerated electron (e−)/hole(h+) pairs, but can also broaden the light responsive spectra, and thereby facilitate the oxygen molecule activation. It also reveals that the photocurrent intensity of the 4% Au/BMO sample is approximately 36-fold larger than that of pure BMO, and the ESR signal intensities of ˙O2− and ˙OH for Au/BMO exhibit an enhancing tendency compared to pure-BMO, demonstrating that SOVs and Au NPs are responsible for the promoted photocatalytic activity of the Bi2MoO6 nanostructure. Furthermore, the band gap position of Au/BMO was determined by employing UV-vis-DRS spectra, VB-XPS and Mott–Schottky plots, thus the enhancement mechanism of oxygen molecule activation is further elucidated.


 Please wait while we load your content...
Please wait while we load your content...