Insight into the branched alcohol formation mechanism on K–ZnCr catalysts from syngas
Abstract
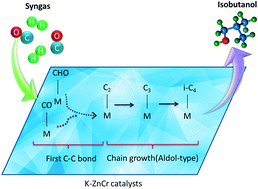
Alcohols produced over K–ZnCr catalysts do not follow the typical Anderson–Schulz–Flory (A–S–F) distribution. Different from traditional linear alcohols, the branched alcohol formation mechanism has not been fully understood to date, especially the formation of the first C–C bond. Herein, a chemical-enrichment method and in situ DRIFTS technique are employed to investigate the formation mechanism of alcohols on K–ZnCr catalysts. The β-addition reaction (aldol-condensation) easily occurs on linear alcohols to form branched alcohols with a methyl group on β-carbon, which is the reason why methanol and isobutanol are the main alcohol products rather than linear alcohols. The adsorbed n-propanol is proved to be the main precursor of isobutanol. Chemical trapping experiment results reveal that formyl species are the C1 intermediates which participate in the synthesis of methanol and higher alcohols. Catalytic performance and in situ DRIFTS results indicate that bicarbonate species are beneficial for the formation of isobutanol, which participates as the precursor of formyl species during the reaction. By comparing the formation ability of alcohols under different kinds of feed gases (CO/H2 and CO2/H2), it is found that the first C–C bond is not formed from two molecules of formyl or CHxO species but from formyl and molecularly adsorbed CO. Once the C2 intermediate is formed, β-addition will react rapidly, leading to the formation of n-propanol and isobutanol successively.


 Please wait while we load your content...
Please wait while we load your content...