One-step green conversion of benzyl bromide to aldehydes on NaOH-modified g-C3N4 with dioxygen under LED visible light†
Abstract
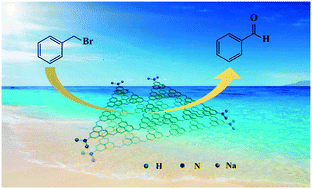
Kornblum oxidation (benzyl bromide to benzaldehyde) can be realized efficiently by NaOH-modified graphitic carbon nitride (g-C3N4). Different from the oxidation reaction carried out at high temperature using DMSO as an oxidant, our reaction is eco-friendly because it is carried out at room temperature under LED light irradiation using molecular oxygen as an oxidant, which avoids the problems of toxicity, pollution and purification. Alkali treatment leads to the surface deprotonation of g-C3N4 to form NaOH-modified g-C3N4 (Na/g-C3N4). The modifications of the electronic structure and surface chemical environment of g-C3N4 facilitate light absorption and formation of ˙O2− and therefore make the conversion of benzyl bromide reach up to 99%. This work demonstrates a green and sustainable route for benzaldehyde production and the superiority of g-C3N4 for organic syntheses.


 Please wait while we load your content...
Please wait while we load your content...