Study of synergistic effect between CuO and CeO2 over CuO@CeO2 core–shell nanocomposites for NH3-SCO†
Abstract
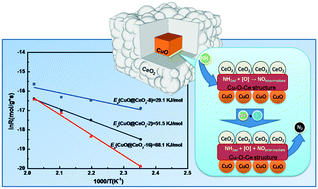
Cerium oxides were orientally introduced into copper oxide phases to improve the catalytic performance of the CuO catalyst for the selective catalytic oxidation of ammonia (NH3-SCO). A series of CuO@CeO2-X (X = 2, 4, 8, and 16) core–shell nanocomposites was fabricated from Cu2O cubic particles via the template-assisted method involving the liquid (Ce(IV))–solid (Cu2O) interfacial reaction and tested as catalysts. Their compositions, structures, catalytic activities and reaction route were studied in detail through HRTEM, XRD, XPS and Raman spectroscopy. The results demonstrated that the synergism between CuO and CeO2 at their interface is enhanced gradually with the formation of the core–shell structure, which facilitates the formation of the Cu–O–Ce structure (solid solution). The CuO@CeO2-8 catalyst with a complete core–shell structure showed the highest catalytic activity, especially at a low temperature (7.0 × 10−2 μmol g−1 s−1 at 180 °C), and the lowest reaction activation energy (Ea = 29.1 J mol−1) among the samples. In addition it was found that the synergistic effect between CuO and CeO2 effectively improves the adsorption and activation of ammonia species from NH3-TPD and NH3 + O2 experiments. Also, more NO intermediate species form on the Cu–O–Ce structure than N2O intermediates, which is conductive to achieve high N2 selectivity following the iSCR route.


 Please wait while we load your content...
Please wait while we load your content...