Influence of carbon doping concentration on photoelectrochemical activity of TiO2 nanotube arrays under water oxidation†
Abstract
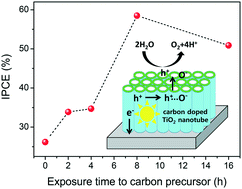
The effect of dopants on photocatalytic activity has been extensively studied because of its potential for the smart design of more energy- and cost-efficient catalytic materials. Here, we investigated the quantitative effects of carbon dopants on TiO2 nanotube arrays for photoelectrochemical (PEC) water splitting. This was enabled by doping different concentrations of interstitial carbon into TiO2 nanotube arrays using thermal treatment with controlled exposure time to the carbon precursor (ethanol vapor). There was no change in the morphology of the nanotube arrays, but the dopant (or defect) levels that were introduced into the TiO2 band gap absorbed visible light (>425 nm) to overcome the relatively large band gap of the TiO2, which only reacts under UV light. We also examined interstitial carbon dopants that form oxygen vacancies, which can suppress the recombination of photogenerated charge carriers by temporarily trapping holes. Those two improvements increased the number of available charge carriers, resulting in enhanced PEC performance. The carbon doped TiO2 nanotube arrays fabricated under eight hours of ethanol exposure exhibited maximal efficiency and photocatalytic activity under simulated solar light.


 Please wait while we load your content...
Please wait while we load your content...