New horizon in C1 chemistry: breaking the selectivity limitation in transformation of syngas and hydrogenation of CO2 into hydrocarbon chemicals and fuels
Abstract
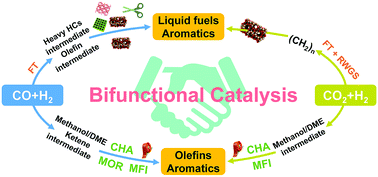
Catalytic transformations of syngas (a mixture of H2 and CO), which is one of the most important C1-chemistry platforms, and CO2, a greenhouse gas released from human industrial activities but also a candidate of abundant carbon feedstock, into chemicals and fuels have attracted much attention in recent years. Fischer–Tropsch (FT) synthesis is a classic route of syngas chemistry, but the product selectivity of FT synthesis is limited by the Anderson–Schulz–Flory (ASF) distribution. The hydrogenation of CO2 into C2+ hydrocarbons involving C–C bond formation encounters similar selectivity limitation. The present article focuses on recent advances in breaking the selectivity limitation by using a reaction coupling strategy for hydrogenation of both CO and CO2 into C2+ hydrocarbons, which include key building-block chemicals, such as lower (C2–C4) olefins and aromatics, and liquid fuels, such as gasoline (C5–C11 hydrocarbons), jet fuel (C8–C16 hydrocarbons) and diesel fuel (C10–C20 hydrocarbons). The design and development of novel bifunctional or multifunctional catalysts, which are composed of metal, metal carbide or metal oxide nanoparticles and zeolites, for hydrogenation of CO and CO2 to C2+ hydrocarbons beyond FT synthesis will be reviewed. The key factors in controlling catalytic performances, such as the catalyst component, the acidity and mesoporosity of the zeolite and the proximity between the metal/metal carbide/metal oxide and zeolite, will be analysed to provide insights for designing efficient bifunctional or multifunctional catalysts. The reaction mechanism, in particular the activation of CO and CO2, the reaction pathway and the reaction intermediate, will be discussed to provide a deep understanding of the chemistry of the new C1 chemistry routes beyond FT synthesis.
- This article is part of the themed collection: Celebrating a Century of Excellency in Chemistry at Xiamen University


 Please wait while we load your content...
Please wait while we load your content...