Solvation properties of protic ionic liquids and molecular solvents†
Abstract
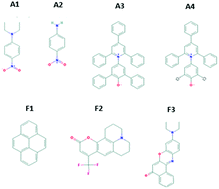
Ionic liquids (ILs) are highly tailorable solvents with many potential applications. Knowledge about their solvation properties is highly beneficial in the utilization of ILs for specific tasks, though for many ILs this is currently unknown. In this study, we have investigated the solvation properties of 12 protic ionic liquids (PILs) and 9 molecular solvents based on the Kamlet–Abboud–Taft′ (KAT) multi-parameter solvation scales. The KAT parameters, which are dipolarity/polarizability (π*), HBD acidity (α), HBA basicity (β), and the electronic transition energy (ET) were first obtained for the molecular solvents with an extensive set of 11 solvatochromic probe dye molecules. Based on these results the dyes which exhibited the highest sensitivities to polarity changes, and had the greatest chemical stability, were used to determine the KAT parameters of 12 PILs which contained alkyl-, dialkyl-, alkanol-, or dialkanolammonium cations paired with nitrate, formate or acetate anions. Solvation parameters were also obtained for the PILs using the three fluorescent probes pyrene, Coumarin 153 and Nile red for comparison. The PILs containing nitrate anions showed the greatest polarity, polarizability and HBD acidity followed by those containing formates and acetates. Almost all the PILs were found to have solvation properties comparable to water and single short chain alcohols like methanol and ethanol. The relative order of the IL polarities was similar for the solvatochromic and fluorescent probes. Through this study, in addition to the well-known distinct solvent properties of alkylammonium cation PILs, the high solvation capability of these PILs has been explicitly shown, which makes this class of ILs desirable for solvent-sensitive applications which require high polarity and H bonding ability.
- This article is part of the themed collection: 2019 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...