Selective conformational control by excitation of NH imino vibrational antennas†
Abstract
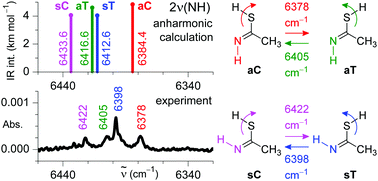
An imino group was used for the first time as a vibrational antenna to manipulate molecular conformations. Imino-thiol isomers of thioacetamide were generated upon UV-irradiation of its amino-thione tautomer isolated in argon matrices at 11 K. Selective and reversible conformational isomerizations were induced by narrowband near-IR irradiation tuned at the frequencies of the 2ν(NH) first stretching overtone of each imino-thiol isomer. The conformational isomerization concerns the change in the orientation of a remote –SH group, while the orientation of the imino (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NH) group remains the same. Supported by quantum chemical anharmonic computations, this allowed for a reliable, isomer-selective vibrational assignment of the four imino-thiol isomers extending now over the full mid-IR and near-IR ranges. It was found that the experimental IR intensities of the 2ν(NH) first stretching overtones (computed 4–5 km mol−1) of the imino-thiol forms are comparable to those of the ν(NH) stretching fundamentals (computed 2–4 km mol−1). This is the first time such a phenomenon is reported for an imine molecule. The kinetics of conformational isomerization was monitored in situ, indicating that the irradiation-induced processes are significantly faster than the tunneling-driven spontaneous cis–trans rotamerization of the –SH group. Quantum yields for the rotamerizations of the –SH group resulting from the vibrational excitation of a remote –NH group were estimated and found to be comparable to those observed for matrix-isolated carboxylic acids and amino acids, where conformational changes of the –OH group were induced by the direct vibrational excitation of 2ν(OH) first stretching overtones.
NH) group remains the same. Supported by quantum chemical anharmonic computations, this allowed for a reliable, isomer-selective vibrational assignment of the four imino-thiol isomers extending now over the full mid-IR and near-IR ranges. It was found that the experimental IR intensities of the 2ν(NH) first stretching overtones (computed 4–5 km mol−1) of the imino-thiol forms are comparable to those of the ν(NH) stretching fundamentals (computed 2–4 km mol−1). This is the first time such a phenomenon is reported for an imine molecule. The kinetics of conformational isomerization was monitored in situ, indicating that the irradiation-induced processes are significantly faster than the tunneling-driven spontaneous cis–trans rotamerization of the –SH group. Quantum yields for the rotamerizations of the –SH group resulting from the vibrational excitation of a remote –NH group were estimated and found to be comparable to those observed for matrix-isolated carboxylic acids and amino acids, where conformational changes of the –OH group were induced by the direct vibrational excitation of 2ν(OH) first stretching overtones.
- This article is part of the themed collection: 2019 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...