Understanding the intramolecular vibrational energy transfer and structural dynamics of anionic ligands in a photo-catalytic CO2 reduction catalyst†
Abstract
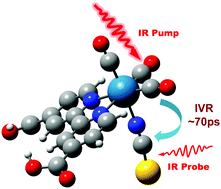
The knowledge of intramolecular vibrational energy redistribution (IVR) and structural dynamics of rhenium photo-catalysts is essential for understanding the mechanism of the photo-catalytic process of CO2 reduction. In this study, the rhenium compound Re(dcbyp)(CO)3NCS (Re-NCS), which served as a model CO2 reduction catalyst, was investigated using two dimensional infrared (2D IR) spectroscopy. The vibrational relaxation dynamics and rotational dynamics of Re-NCS were measured by monitoring both the CO and NCS vibrational modes. The rotational dynamics measurement of the CO vibrational stretch shows a single exponential decay with a time constant of 140 ± 10 ps. In contrast, a bi-exponential decay is needed to describe the rotational dynamics of the NCS stretching mode with time constants of 1.5 ± 0.3 ps and 189 ± 15 ps. The 2D IR experiment indicated that the carbonyl CO vibrational modes in Re-NCS are strongly coupled. Furthermore, the intramolecular vibrational energy transfer between CO and NCS stretching modes was observed and analyzed based on an energy exchange model. The energy down flowing transfer from CN to CO stretching mode was determined using time constants of 50 ps. The relatively slow intramolecular vibrational energy transfer rate suggests that there is a weak coupling between CO and NCS ligands. Further theoretical calculation showed that the coupling strength between CO and CN is relatively weak and is about 5–6 times smaller than the coupling strength between the CO vibrational modes in Re-NCS. The distinct structural dynamics of the NCS ligand in Re-NCS presented in this study should provide a fundamental understanding of the role of an anionic ligand in rhenium photo-catalysts, which is believed to play an important role in the photo-catalytic reduction of CO2.


 Please wait while we load your content...
Please wait while we load your content...