Chemistry of iron nitrate-based precursor solutions for spray-flame synthesis
Abstract
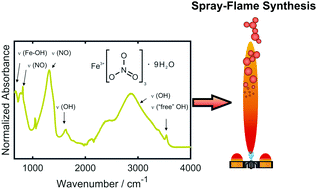
Understanding the chemistry of iron-based metal–organic precursor solutions for spray-flame synthesis is a key step to developing inexpensive and large scale applications for gas-phase synthesized, nano-sized iron oxide particles. Owing to the large variety of available organic solvents and iron compounds, the choice of a suitable precursor-solvent pair is challenging. Systematic investigations of the precursor chemistry of iron-based systems are currently not available. This work aims at filling this gap by providing a detailed spectroscopic analysis of mixtures containing iron(III) nitrate nonahydrate and alkyl alcohols (C2–4). Moreover, the impact of adding 2-ethylhexanoic acid is explored. The FTIR spectra reveal the formation of carboxylates and allow deriving information about the coordination of the metal-carboxylate complexes. The stability of the precursor solutions is investigated by monitoring precipitation phenomena and turbidity. Furthermore, gas chromatography is employed to provide additional information on oxidation products and esters as well as to aid the interpretation of the FTIR data. It is found that the formation of esters has an enhancing effect on iron sorption and, thus, it promotes precursor stability.


 Please wait while we load your content...
Please wait while we load your content...