Theoretical research on the oxidation mechanism of doped carbon based catalysts for oxygen reduction reaction†
Abstract
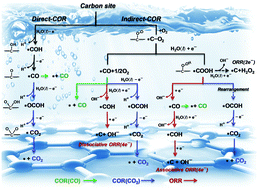
To understand the essential reasons of poor durability and rapid initial performance loss of heteroatom doped graphene catalysts during the electrochemical oxygen reduction reaction (ORR) process, it is necessary to explore the detailed mechanism of carbon active site oxidation reaction (COR) at different electrode potentials, as it may greatly influence the ORR activity. Herein, density functional theory (DFT) calculation is used to investigate all possible COR mechanisms, including Direct-COR and Indirect-COR, on four typical doped-graphene, and understand the competing relation between COR and ORR from a thermodynamic point of view. Our systematic calculations found that the Direct-COR is affected directly by the structural stability of doped-graphene relative to pure graphite, and the Indirect-COR can be accelerated largely by the ORR process due to the ORR intermediate, such as O and OOH. The competition relation between COR and ORR is mainly influenced by the interaction between the doped-graphene and reaction species, stability of doped-structure, ORR mechanism, and electrode potential. For COR, the partial oxidation of doped-graphene is the dominant oxidation reaction compared to complete oxidation in the ORR potential range. More importantly, both partial and complete oxidation of doped-graphene can remarkably depress the ORR activity. Hence, COR should be one of the major contributors to the rapid initial performance loss of carbon based catalysts in stability testing. Our results provide a comprehensive and deep understanding of the oxidation of carbon active sites on doped-graphene surfaces and can guide the design of more robust doped-carbon based catalysts.


 Please wait while we load your content...
Please wait while we load your content...