Promotional effect of in situ generated hydroxyl on olefin selectivity of Co-catalyzed Fischer–Tropsch synthesis†
Abstract
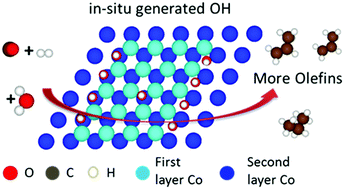
The understanding of the water effect on olefin selectivity in Fischer–Tropsch synthesis (FTS) is limited by the complexity of the reaction network. Herein, we employ propene hydrogenation as a model reaction to isolate the water effect on olefin adsorption and hydrogenation from the complex reaction of FTS. It is clearly observed that the added water inhibits the activity of propene hydrogenation on two cobalt catalysts supported on high-surface-area alumina (HAS Al2O3) and low-surface-area alumina (LSA Al2O3), respectively. The inhibiting effect is much stronger for Co/HSA Al2O3. DFT investigation demonstrates that the in situ generated OH, rather than H2O and O, impedes the adsorption of propene and thus decreases the activity of propene hydrogenation. The suppressive effect of OH on propene adsorption is attributed to the downshift of the d-band center and the Bader charge of the catalyst surface. The DFT-based kinetic analysis finds that the higher site coverage of OH results in the more pronounced negative effect on propene hydrogenation. Furthermore, the theory of OH-induced weak olefin adsorption and low olefin hydrogenation activity could rationalize the enhancement effect of water on the olefin selectivity and the particle size dependence of the water effect in FTS. The insights obtained here may inspire researchers to optimize olefin selectivity by manipulating the electronic properties of catalysts with hydroxyl species.
- This article is part of the themed collection: 2019 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...