Interpenetrating graphene network bct-C40: a promising anode material for Li ion batteries
Abstract
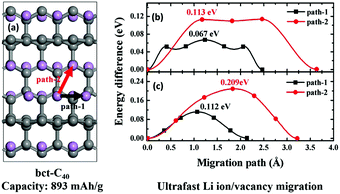
The stable sp2-C atoms in graphite enable its excellent structural and electrochemical stability as an anode material for Li-ion battery applications, while the limited Li-storage capacity of graphite also originates from the sp2 hybridization. Herein, from first-principles calculations, we show that a synergistic effect of sp2 and sp3 hybridized C atoms can substantially enhance the Li-storage performance in carbon-based anodes, using bct-C40 as an example, which is constructed with interconnected graphene layers (sp2 hybridized C atoms) and the connecting points are composed of sp3-C atoms. Charge transfer from sp2-C atoms to sp3-C atoms has been found, leading to unoccupied electronic states forming around the Fermi level. Furthermore, we found that the unoccupied electronic states are contributed by the pz orbital of the sp2-C atoms, resulting in stronger interactions between C atoms and intercalated Li atoms. As a result, the Li intercalation concentration in bct-C40 can reach as high as LiC2.5 (corresponding to a capacity of 893 mA h g−1), much higher than that of LiC6 in graphite (372 mA h g−1). Furthermore, bct-C40 inherits good structural and electrochemical stability, a metallic electronic structure, and low Li-ion migration energy barriers (0.067–0.112 eV) from the sp2 hybridized graphene structures, therefore very good Li-storage performance is expected, indicating that bct-C40 can be used as a high-performance anode material for lithium ion batteries. Our study provides new insights into the functionality of sp2- and sp3-C atoms in carbon-based anode materials and is helpful for the designing of new carbon-based anodes.


 Please wait while we load your content...
Please wait while we load your content...