Conjugation-broken thiophene-based electropolymerized polymers with well-defined structures: effect of conjugation lengths on electrochromic properties†
Abstract
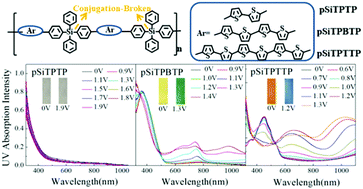
A series of monomers containing tetraphenylsilane connected to different thiophenes such as thiophene, bithiophene and terthiophene were designed and synthesized and were further used to prepare the corresponding polymers via electrochemical polymerization (pSiTPTP, pSiTPBTP and pSiTPTTP). From the polymers, the effective conjugate elements were well defined as bithiophene, quaterthiophene and sexithiophene because the sp3 Si atom can block the conjugation between the thiophene units in the polymer backbone. The spectroelectrochemical results indicated that pSiTPTP is incapable of electrochromism, which may be attributed to the insufficient conjugation length of the independent bithiophene. In contrast, both pSiTPBTP and pSiTPTTP exhibited obvious electrochromic properties and furthermore, pSiTPTTP displayed a shorter switching time and better stability. Such different electrochemical behaviors can be ascribed to the looser stacking structure and lower oxidation potential of pSiTPTTP with the independent sexithiophene unit. The EIS measurements also confirmed the lower charge-transfer resistance and higher ion-diffusion rate of pSiTPTTP with the independent sexithiophene unit. Hence, we can conclude that the effects of the electrochromic behavior of the conjugation-broken polythiophene derivatives depend on the increased conjugation length of the thiophene repeating unit, in which the inadequate electrochromism with bithiophene units can change to superior electrochromic properties with increased sexithiophene units.


 Please wait while we load your content...
Please wait while we load your content...