Effects of micro-bubbles on the nucleation and morphology of gas hydrate crystals
Abstract
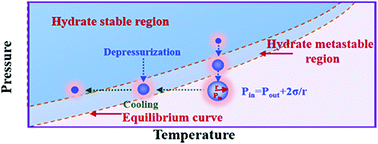
Gas hydrate is usually regarded as a huge potential energy resource that has promising industrial applications in gas separation, storage, and transportation. Previous research studies have shown that a gas hydrate phase transition is mainly controlled by heat and mass transfer, while there are limited works on the mass transfer effects of gas micro-bubbles on hydrate crystallization. In this study, variations in the microscopic morphology of the hydrate crystal growth in a liquid–gas interface were observed using a microscope imaging system. The results indicated that the nucleation of the hydrate first tends to occur at the bubble surface. The cooling rates increased exponentially with the crystal growth rates and played an important role in the morphology of the hydrate crystal growth. In addition, the hydrate crystals tended to grow in the direction of the bubbles affected by the Ostwald ripening effects, which suggested that bubbling was an efficient measure to promote the application of hydrate-based technologies. In turn, reducing the concentration of the bubbles on the surface of the hydrate, inhibiting their generation, and enhancing the process of gas mass transfer in water around the hydrate surface were also conducive to further accelerate the decomposition of the hydrate, which may provide some guidance for the resource exploitation of gas hydrate.
- This article is part of the themed collection: 2019 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...