Understanding of binding energy calibration in XPS of lanthanum oxide by in situ treatment†
Abstract
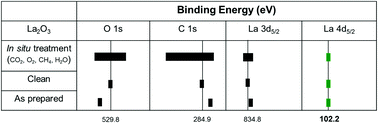
Rare earth oxides have seen increased usage over the years in batteries and catalysts. Due to their unique electronic properties, they are the subject of fundamental and practical interest. However, the complexity in their electronic structures makes unambiguous characterization, such as X-ray photoelectron spectroscopy (XPS), very challenging. Lanthanum oxide (La2O3) has attracted special attention as a promising catalyst for the oxidative coupling of methane (OCM) reaction. In this work, a new and reliable way of XPS calibration is developed by applying various in situ preparations for a nanorod La2O3 catalyst to intentionally form different lanthanum compounds, followed by XPS characterization and corroboration with first principles calculations. To form different compounds, five sample treatments were performed including heating in vacuum and treatment with O2, CH4, CO2, and H2O, which are all relevant to OCM reaction conditions. Adventitious carbon or lattice oxygen, as conventional calibration standard species for energy scale, is only suitable for one or few in situ prepared surfaces. Our results also clearly demonstrate the vital difference between performing the ex situ analysis after exposure of the sample to the atmosphere and the in situ analysis. By carefully comparing the spectra of various photoemission peaks of different compounds, we conclude that the binding energy of 102.2 eV for the La 4d7/2 peak can be used as the internal calibration standard for all considered samples. Furthermore, different oxygen species were unambiguously identified by matching the oxygen 1s binding energies from the in situ measurements and first principles predictions.


 Please wait while we load your content...
Please wait while we load your content...