Electronic structure of CsPbBr3−xClx perovskites: synthesis, experimental characterization, and DFT simulations†
Abstract
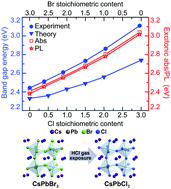
All-inorganic lead halide perovskites of various compositions have emerged as a prospective family of materials for light-emitting devices and photonic applications. However, a comprehensive study of their structural and electronic properties is still missing. Moreover, thin film fabrication of these perovskites comprising heterohalide anions by wet chemistry approaches also remains challenging. Here we fabricate high-quality CsPbBr3−xClx perovskite thin films using a wet chemical method accompanied by a chemical vapor anion exchange procedure, which allows rigorously studying their optical and structural properties at different compositions. Namely, we present both the numerical and experimental studies of the electronic properties of all-inorganic mixed-halide perovskites, showing their optical absorption, excitonic photoluminescence and exciton binding energy, phase, chemical composition, and band structure and the band gap evolution with a gradual change in x in CsPbBr3−xClx. The results reveal that as Cl ions substitute for Br ones in the perovskite crystal lattice the room-temperature phase does not change its orthorhombic symmetry, whereas the energy of the direct electronic transition from the valence to conduction band at the Γ-point increases nonlinearly. By using the experimentally derived nonlinear dependence it is easy to accurately predict the band gap for any CsPbBr3−xClx perovskite thin film consisting of grains with sizes beyond the quantum confinement regime.
- This article is part of the themed collection: 2019 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...