Quantitative determination of the phosphorus environment in lithium aluminosilicate glasses using solid-state NMR techniques
Abstract
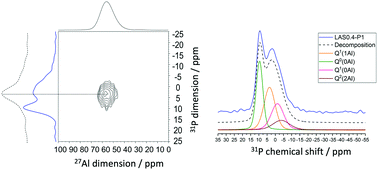
We investigated using solid-state NMR spectroscopy the short-range structural features in lithium aluminosilicate glasses with the addition of P2O5 and considering various Al2O3/Li2O ratios. The phosphorus environment is determined quantitatively using 31P Magic Angle Spinning NMR constrained by results obtained from 31P–27Al Multiple-Quantum Coherence-based NMR techniques. Phosphorus is mainly located as orthophosphate and pyrophosphate species in glasses with a low amount of Al2O3. These depolymerized units disappear with increasing Al2O3 content and a strong affinity of PO4 tetrahedra for aluminum is revealed, which reduces phase separation. The local environments of framework (Si and Al) and charge-balancing (Li) cations are also studied through NMR experiments to assess the influence of P2O5 addition. The Si environment is mostly modified by the presence of P2O5 in glasses containing a low amount of Al2O3, with an increase of Q4Si species in relation to phase separation phenomena observed in these compositions. Conversely, P2O5 addition does not have a significant influence on the 27Al NMR response. 7Li NMR spectra reflect a change in the structural role of Li when P2O5 or Al2O3 is added. The observed structural changes can be rationalized to improve our knowledge of the structure–property relationships, focusing, in particular, on phase separation and nucleation/crystallization processes that are strongly affected by the presence of P and the evolution of its local environment with composition.


 Please wait while we load your content...
Please wait while we load your content...