A reaction energy profile and fragment attributed molecular system energy change (FAMSEC)-based protocol designed to uncover reaction mechanisms: a case study of the proline-catalysed aldol reaction†
Abstract
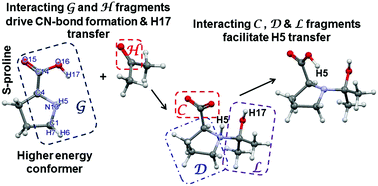
A REP-FAMSEC (reaction energy profile-fragment attributed molecular system energy change) protocol designed to explain each consecutive energy change along the reaction pathway is reported. It mainly explores interactions between meaningful polyatomic fragments of a molecular system and, by quantifying energetic contributions, pin-points fragments (atoms) leading to or opposing a chemical change. Its usefulness is tested, as a case study, on the proline-catalysed aldol reaction for which a number of mechanisms have been debated for over four decades. The relative stability of S-proline conformers, their catalytic (in)activity and the superior affinity of the higher energy conformer to acetone is fully explained at atomic and molecular fragment levels, but still appealing to general chemist knowledge. We found that (i) contrary to the generally accepted view, CN-bond formation cannot be explained by the Nδ−, Cδ+ atom pair, but rather by the O-atom of acetone and its strongest inter-molecular attractive interactions with the N-atom as well as the C-atom of the COO group of proline (at this initial stage the lower energy conformer of proline is eliminated) and (ii) the following ‘first’ H-transfer from N to O atoms of the proline moiety is nearly energy-free even though initially the H-atom interacts three times stronger with the N- than O-atom; a full explanation of this phenomenon is provided.


 Please wait while we load your content...
Please wait while we load your content...