Excited state structural dynamics of 4-cyano-4′-hydroxystilbene: deciphering the signatures of proton-coupled electron transfer using ultrafast Raman loss spectroscopy†‡
Abstract
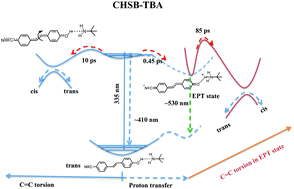
The photo-initiated proton-coupled electron transfer (PCET) process plays a crucial role in the context of light harvesting in various biological and chemical systems. Molecular model systems are typically employed to understand the mechanisms underlying the functioning of complex biological systems. Some molecular dyads based on the PCET property have been particularly designed to achieve efficient sunlight-to-fuel production. Organic photoacids are potential sources for such applications since they exhibit an enhancement in their acidity upon photoexcitation, facilitating the mimicking of some of the biological processes. p-Hydroxybenzylideneimidazolinone (p-HBI), an organic photoacid, is a key chromophore in green fluorescent protein, which exhibits green emission due to excited state proton transfer. Herein, we investigate the structural changes and dynamics of 4-cyano-4′-hydroxystilbene (CHSB), an analogue of p-HBI, in the presence of an external base, t-butylamine (TBA), using the techniques of ultrafast transient absorption, emission and ultrafast Raman loss spectroscopy. Femtosecond fluorescence up-conversion measurements of the CHSB–TBA adduct reveal a precursor–successor relationship between the ∼420 and ∼530 nm emission bands, which implies that the adduct evolves predominantly through an electron–proton transferred state. Further, Raman measurements show a clear distinction in the dynamics of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretch of CHSB in the presence and absence of TBA in terms of the amplitude growth (0.45 ps vs. instantaneous) and the central frequency (1584 vs. 1523 cm−1).
C stretch of CHSB in the presence and absence of TBA in terms of the amplitude growth (0.45 ps vs. instantaneous) and the central frequency (1584 vs. 1523 cm−1).


 Please wait while we load your content...
Please wait while we load your content...