Thermodynamic properties and lattice dynamics investigation of LuB2C: experiment and ab initio calculations†
Abstract
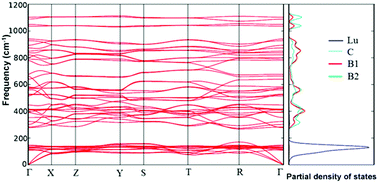
A sample of lutetium carboboride LuB2C was synthesized from a mixture of lutetium hydride, boron and carbon by annealing in argon. The temperature dependence of the heat capacity Cp(T) (2–300 K) and lattice parameters a(T), b(T), and c(T) (5–300 K) of the carboboride was experimentally determined. The experimental values of the heat capacity were fitted with the approximation Cp(T) = aT + ΣCD + CE + CTLS(T). Here the first term is the electronic contribution, the second is the sum of the Debye components, the third is the Einstein contribution, and the fourth is the contribution to the heat capacity due to the vibrations of the two-level systems which are formed in the Lu-subsystem due to the asymmetry of the B–C atomic arrangement around the Lu3+-ions and, as a consequence, the possible transition of the lutetium atoms between spatially close, but energetically non-equivalent positions. A strong anisotropy of the thermal expansion of the carboboride was revealed. Along the c axis the coefficient of thermal expansion monotonically increases; in the basal plane, the expansion is practically not observed. The temperature dependence of the unit cell volume Vu(T) has been analyzed in the Debye–Einstein approximation taking into account the electronic contribution and effect of two-level systems. The values of the Gruneisen parameters corresponding to different modes of the phonon spectrum of the carboboride have been determined. The frequencies of the lattice vibrations, determined in a Raman scattering experiment, are in satisfactory agreement with the parameters obtained from Cp(T) using the Debye–Einstein approximation. Using ab initio band theory methods and an exchange–correlation functional in the PBE form in the VASP package, it was established that the total energies of these two crystal structures differ by no more than 0.01 eV f.u.−1. Calculations of the thermodynamic properties of LuB2C yielded similar results for orthorhombic and tetragonal phases of the carboboride.


 Please wait while we load your content...
Please wait while we load your content...