Hydrogenated polycyclic aromatic hydrocarbons (HnPAHs) as catalysts for hydrogenation reactions in the interstellar medium: a quantum chemical model
Abstract
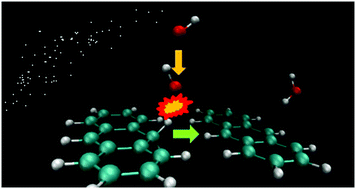
The sticking of H atoms onto dust grains and large hydrocarbon molecules has received considerable attention because it is thought to govern the formation of H2 and other H-containing molecules in the interstellar medium. Using the density functional theory (DFT) approximation, we have investigated the capacity of neutral hydrogenated polycyclic aromatic hydrocarbons (HnPAH) to catalyze simple hydrogenation reactions by acting as a source of atomic hydrogen. In particular, the interaction of OH and CO with H1-anthracene (singly hydrogenated) and H14-anthracene (fully hydrogenated) to form H2O and HCO was modeled following the Eley–Rideal mechanism. In this process, a hydrogen atom is abstracted from the HnPAH molecule forming the corresponding hydrogenated compound. The results were compared to the most known case of the HnPAH-catalyzed formation of H2. It was observed that whereas H2 is formed by overcoming activation barriers of approximately 0.02 and 0.10 eV with H1-anthracene and H14-anthracene, respectively, H2O is produced in a barrierless fashion with both hydrocarbon molecules. The production of HCO was found to be a highly unfavorable process (with activation barriers of 0.73 eV and 3.13 eV for H1- and H14-anthracene, respectively). Complementary calculations performed using the rest of the Hn-anthracene molecules (from 2 to 13 extra H atoms) showed that in all the cases the reaction with OH is barrierless as well. This efficient mechanism could therefore be a possible route for water formation in the cold interstellar medium.
- This article is part of the themed collection: Celebrating Latin American Talent in Chemistry


 Please wait while we load your content...
Please wait while we load your content...