Atomistic insight into the inhibition mechanisms of suppressors of cytokine signaling on Janus kinase†
Abstract
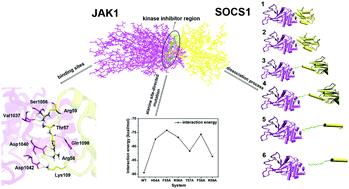
Suppressors of cytokine signaling (SOCS) act as negative feedback regulators of the Janus kinase/signal transducer (JAK–STAT) signaling pathway by inhibiting the activity of JAK kinase. The kinase inhibitory region (KIR) of SOCS1 targets the substrate binding groove of JAK with high specificity, as demonstrated by significantly higher IC50 following the mutation of any of residue. To gain a greater understanding of the mechanisms of the inhibition of SOCS1 for JAK1, the binding mode, binding free energy decomposition, and desorption mechanism of JAK–SOCS1 complexes as well as a number of mutant systems were identified by extensive molecular dynamics (MD) simulations and the constant pulling velocity (PCV) method. Electrostatic interactions were identified for their contribution to protein–protein binding, which drove interactions between JAK1 and SOCS1. The polar residues Arg56, Arg59, and Asp105 of SOCS1 and Asp1042 and Asp1040 of JAK1 were key components in the binding, and electrostatic interactions of the side chains were prominent. The binding free energies of the six mutant proteins were lower when compared with those of the control proteins, and the side chain interactions were weakened. The residue Asp1040 played a crucial role in KIR close to the binding groove of JAK1. Moreover, salt bridges contributed significantly to JAK1 and SOCS1 binding and cleavage processes. The study presented herein provides a comprehensive understanding of the thermodynamic and dynamic processes of SOCS1 and JAK1 binding that will contribute meaningfully to the design of future studies related to peptide inhibitors based on SOCS1.


 Please wait while we load your content...
Please wait while we load your content...