Modulating amyloid fibrillation in a minimalist model peptide by intermolecular disulfide chemical reduction†
Abstract
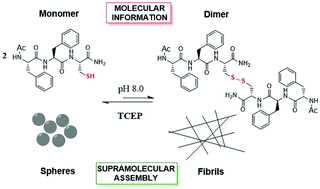
Peptide structural transformation and aggregation is associated with a large number of outsider aetiology diseases, and it is intrinsically linked to amyloid peptide aggregation. Diphenylalanine self-assembled structures are used as robust minimalist beta amyloids not only to elucidate protein aggregation but also to generate hydrogels. Herein, we employed a neutral model peptide Ac-Phe-Phe-Cys-NH2 (Ac-FFC-NH2) to elucidate the role of intermolecular disulfide bonds in protein fibrillation. The Ac-FFC-NH2 peptide initially self-assembles into nanospheres that evolve to amyloid type fibrils under mild oxidative conditions. Incubation of the peptide in the presence of the chemical reduction agent TCEP inhibits the formation of the fibrils, detecting only spherical nanostructures with no secondary structure. Importantly, we triggered the transformation of the preformed linear straight amyloid fibrils to non-fibrillar structures by TCEP treatment. Under this condition, the amyloid bundles are transformed into rings, which evolve to a new spherical microstructure. We showed that the chemical reduction of intermolecular S–S in internal amyloid sequences might favour the off-path intermediates of amyloid fibril growth, even when the fibrils are formed. Our findings demonstrated that in internal amyloid sequences, the formation of intermolecular S–S promotes the formation of amyloid type fibrils; meanwhile, its reduction stabilises non-fibrillar structures. Altogether, this work provides fundamental understanding at the molecular and supramolecular level, thus facilitating the rational design of therapeutic tools for protein aggregation diseases.


 Please wait while we load your content...
Please wait while we load your content...