Experimental and first-principles DFT studies on the band gap behaviours of microsized and nanosized Zn(1−x)MnxO materials
Abstract
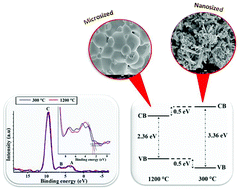
In this study, nano- and microsized zinc oxide (ZnO) materials were doped with different manganese (Mn) contents (1–5 mol%) via a simple sol–gel method. The structural, morphological, optical and chemical environments of the materials were investigated using X-ray diffraction (XRD), field emission scanning electron microscopy (FESEM), energy dispersive X-ray spectroscopy (EDX), UV-visible spectroscopy (UV-vis) and X-ray photoelectron spectroscopy (XPS). XRD results revealed that all synthesised materials were pure and single phased with a hexagonal wurtzite structure of ZnO. However, at a low annealing temperature, a nanorod-like shape can be obtained for all Zn(1−x)MnxO materials. In addition, EDX spectra confirmed the presence of Mn in the ZnO lattice and the atomic percentage was nearly equal to the calculated stoichiometry. UV-vis spectroscopy further revealed that materials in nano size exhibited band gap widening with an increase of the Mn content in the ZnO lattice. In contrast, micron state materials exhibited band gap narrowing with increasing Mn content up to 3% and then begin to widen when Mn > 3%. This is because the band gaps of these materials are affected by the dimensions of the crystals and the Mn content in the materials. Furthermore, XPS results revealed the existence of multiple states of Mn in all synthesised materials. By combining the information obtained from UV-vis and the XPS valence band, shifting in the valence band maximum (VBM) and conduction band minimum (CBM) was observed. Based on XPS results, the calculation of density functional theory studies revealed that the presence of Mn2+, Mn3+, and Mn4+ ions in the materials influences the band gap changes. It was also revealed that the nanosized Zn0.99Mn0.01O exhibited a higher photocatalytic activity than the other samples for degrading methylene blue (MB) dyes, owing to its smallest crystallite size.


 Please wait while we load your content...
Please wait while we load your content...