Molecular mechanisms of pore formation and membrane disruption by the antimicrobial lantibiotic peptide Mutacin 1140†
Abstract
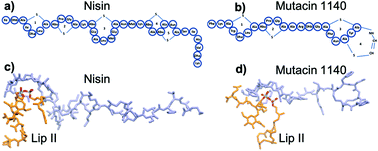
The emergence of antibiotic-resistance is a major concern to global human health and identification of novel antibiotics is critical to mitigate the threat. Mutacin 1140 (MU1140) is a promising antimicrobial lanthipeptide and is effective against Gram-positive bacteria. Like nisin, MU1140 targets and sequesters lipid II and interferes with its function, which results in the inhibition of bacterial cell wall synthesis, and leads to bacteria cell lysis. MU1140 contains a structurally similar thioether cage for binding the lipid II pyrophosphate as for nisin. In addition to lipid II binding, nisin is known to form membrane pores. Membrane pore formation and membrane disruption is a common mode of action for many antimicrobial peptides, including gallidermin, a lantibiotic peptide with similar structural features as MU1140. However, whether and how MU1140 and its variants can form permeable membrane pores remains to be demonstrated. In this work, we explored the potential mechanisms of membrane pore formation by performing molecular simulations of the MU1140–lipid II complex in the bacterial membrane. Our results suggest that MU1140–lipid II complexes are able to form water permeating membrane pores. We find that a single chain of MU1140 complexed with lipid II in the transmembrane region can permeate water molecules across the membrane via a single-file water transport mechanism. The ordering of the water molecules in the single-file chain region as well as the diffusion behavior is similar to those observed in other biological water channels. Multiple complexes of MU1140–lipid II in the membrane showed enhanced permeability for the water molecules, as well as a noticeable membrane distortion and lipid relocation, suggesting that a higher concentration of MU1140 assembly in the membrane can cause significant disruption of the bacterial membrane. These investigations provide an atomistic level insight into a novel mode of action for MU1140 that can be exploited to develop optimized peptide variants with improved antimicrobial properties.


 Please wait while we load your content...
Please wait while we load your content...