Photochromic reaction in 3H-naphthopyrans studied by vibrational spectroscopy and quantum chemical calculations†
Abstract
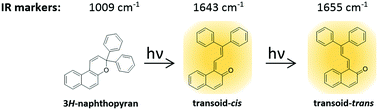
Structural details on the species involved in the photochromic reaction of 3H-naphthopyrans in solution have been formerly determined using NMR spectroscopy. Herein we show that at room temperature time-resolved FT-IR spectroscopy is a simple and efficient tool for structural characterization of colored species generated upon continuous UV light irradiation of the model compound 3H-naphthopyran: 3,3-diphenyl-3H-naphtho[2,1-b]pyran. In solution and in the polymer matrix phase, a colored species transoid-cis is formed after a single-photon excitation process, while transoid-trans is a secondary long-lived photoproduct generated after two-step excitation involving two photons. Understanding the reaction mechanism leading to long-lived colored species can help with the design of new 3H-naphthopyran derivatives structurally optimized for making a photochromic reaction free from transoid-trans products, which is often important for applications. Ab initio calculations show that photoinduced ring-opening followed by isomerization occurs on a multidimensional potential-energy surface. The barriers separating the considered isomeric forms, both in the ground and in the excited state, help to interpret the step-by-step dynamics of the photoprocesses. The system is composed of a variety of ground state equilibrium forms. Each of them is characterized by fast excited-state deactivation pathways which may drive the system through different conical intersection regions.


 Please wait while we load your content...
Please wait while we load your content...