Intermolecular dynamics of ultraconfined interlayer water in tobermorite: influence on mechanical performance
Abstract
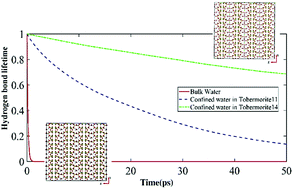
Ultraconfined interlayer water within the tobermorite molecular structure is responsible for changes in the uniaxial tensile and compressive response of the family of tobermorites: 9, 11 and 14 Å. These confined interlayer water molecules are engaged in solvation of cations and anions within the tobermorite structure, which has been demonstrated through the intermolecular vibrational spectra and hydrogen bond lifetime of the water molecules. This study demonstrates that instead of ionization of water molecules (as proposed in an earlier study), breaking of hydrogen bonds of water is more plausible leading to solvation of ions within the molecular structure of tobermorite. A schematic of the coordinate covalent bonds between the water molecules and the cations and anions of the tobermorite structure has been proposed in this study.


 Please wait while we load your content...
Please wait while we load your content...