Tuning the liquid-phase exfoliation of arsenic nanosheets by interaction with various solvents†
Abstract
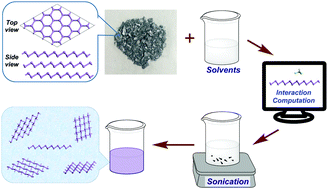
The interaction between monolayered arsenene and fourteen kinds of solvents is found to be correlated with the extent of charge transfer from arsenene to the solvents by density functional theory calculations. Among them, three kinds of aprotic solvents (cyclohexane, tetrahydrofuran and chloroform), representing different adsorbability with arsenene, were selected to exfoliate bulk arsenic crystals into nanosheets in experiments. The as-prepared concentrations of the three dispersions vary monotonically with the calculated adsorption energies and charge transfer per contact area. Transmission electron microscopy (TEM) characterization and size distribution analyses manifested that the lateral size distributions of the exfoliated arsenic nanosheets ranged from 100 nm to 1050 nm. This strategy shows the potential usage in liquid-phase exfoliation of multi-layered materials due to the interaction effects between the solvents and the surface of the materials.


 Please wait while we load your content...
Please wait while we load your content...