Sudden polarization and zwitterion formation as a pseudo-Jahn–Teller effect: a new insight into the photochemistry of alkenes†
Abstract
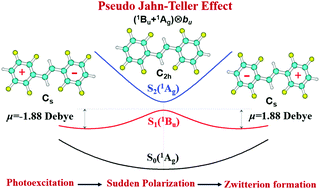
We show that the intermediates of photochemical reactions—sudden polarization and zwitterion formations—are consequences of the pseudo-Jahn–Teller effect (PJTE), which facilitates a better understanding, rationalization, prediction, and manipulation of the corresponding chemical and biological processes. This statement is illustrated on a series of alkenes, including ethylene, butadiene, diallyls, stilbene, and ring-halogenated stilbenes, but the formulated methodology and results are applicable to any photochemical reaction controlled by sudden polarization (and can be extended to other chemical reactions with short-lived intermediates). All these molecules have many polar modes, significantly complicating the PJTE multimode problem. We resolved this by introducing the “zwitterion mode,” which reproduces the integrated effect of all the polar modes and their relative activities by involving ab initio calculated energy and dipole moment profiles, thereby revealing the contribution of the separate parts of the molecular structure to the process of sudden polarization and reactivity. Among the other details, the PJTE analysis shows, for instance, why only ring-fluorinated stilbene exhibits the sudden polarization effect, whereas Cl-stilbene, Br-stilbene, I-stilbene, and planar stilbene exhibit no significant photo-polarization. The possible manipulations of the process are also mentioned. With the example of glycine, we also show that charge separation and zwitterion formation via the PJTE may take place not only as a result of photoexcitation, but also under other external perturbations, e.g., interaction with a polarizable environment. The importance of the PJTE treatment of the problem is also in its direct application to unstable intermediates of chemical reactions, where electronic structure calculations do not converge in the geometry optimization part.


 Please wait while we load your content...
Please wait while we load your content...