Ionic transport in highly concentrated lithium bis(fluorosulfonyl)amide electrolytes with keto ester solvents: structural implications for ion hopping conduction in liquid electrolytes†
Abstract
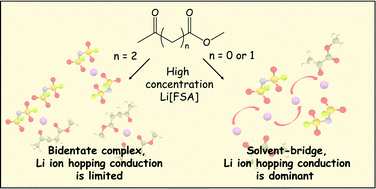
Recent studies have suggested that a Li ion hopping or ligand- or anion-exchange mechanism is largely involved in Li ion conduction of highly concentrated liquid electrolytes. To understand the determining factors for the Li ion hopping/exchange dominant conduction in such liquid systems, ionic diffusion behavior and Li ion coordination structures of concentrated liquid electrolytes composed of lithium bis(fluorosulfonyl)amide (Li[FSA]) and keto ester solvents with two carbonyl coordinating sites of increasing intramolecular distance (methyl pyruvate (MP), methyl acetoacetate (MA), and methyl levulinate (ML)) were studied. Diffusivity measurements of MP- and MA-based concentrated electrolytes showed faster Li ion diffusion than the solvent and FSA anion, demonstrating that Li ion diffusion was dominated by the Li ion hopping/exchange mechanism. A solvent-bridged, chain-like Li ion coordination structure and highly aggregated ion pairs (AGGs) or ionic clusters e.g. Lix[FSA]y(y−x)− forming in the electrolytes were shown to contribute to Li ion hopping conduction. By contrast, ML, with greater intramolecular distance between the carbonyl moieties, is more prone to form a bidentate complex with a Li cation, which increased the contribution of the vehicle mechanism to Li ion diffusion even though similar AGGs and ionic clusters were also observed. The clear correlation between the unusual Li ion diffusion and the solvent-bridged, chain-like structure provides an important insight into the design principles for fast Li ion conducting liquid electrolytes that would enable Li ion transport decoupled from viscosity-controlled mass transfer processes.


 Please wait while we load your content...
Please wait while we load your content...